Recombinant Blood Group Antigens (rBGA)
For the specification of irregular anti-erythrocytic antibodies
The detection of antibodies to blood group antigens is crucial in pre and posttransfusion testing to allow for adequate blood supply of patients requiring blood transfusions. In cases when e.g. antibody mixtures, autoantibodies or alloantibodies to high-prevalence blood group antigens are present, the clear identification could be problematic due to unspecific positive (false positive) or overlapping reactions in antibody screening. Recombinant blood group antigens (rBGA) by imusyn can specifically inhibit antibodies and therefore minimize the risk of incompatible blood transfusions.

Your contact
Dr. Sabrina König
Phone: +49 6173 6079 30
E-mail: s.koenig@inno-train.de
Downloads
Flyer rBGA
Method
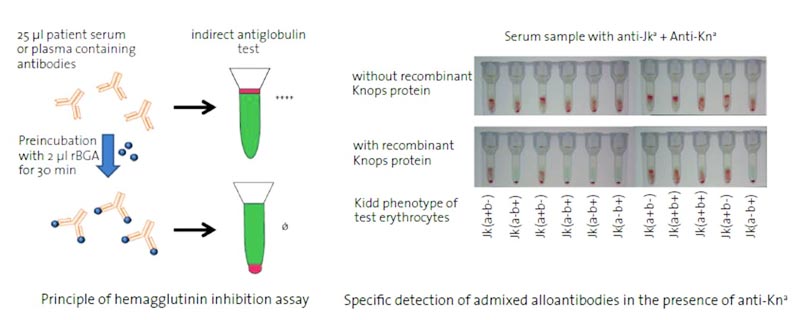
- Preincubation of 2 μl rBGA with 25 μl patient serum: neutralization of specific red cell antibodies
- Serum can be used for indirect antiglobulin test with conventional gel card systems (Grifols DG® Gel Coombs, Bio-Rad ID-Card LISS/Coombs)
rBGA Features & Advantages
Simple test procedure
Direct antibody identification
Antibody detection and identification in one step
Simple and fast detection and identification of alloantibodies to high-prevalence blood group antigens
Better resolution of antibody mixtures
Neutralization of clinically insignificant antibodies to high-prevalence antigens in pretransfusion cross-matching
Faster and safer treatment of immunized patients
Easy to implement in routine serology
Recombinant Blood Group Antigens*
| Item number | Name | Description |
|---|---|---|
| 004 010 001 | Chido(a) | vial à 300 µl |
| 004 010 003 | CROM/DAF | vial à 300 µl |
| 004 010 004 | Dombrock(a) | vial à 300 µl |
| 004 010 005 | Dombrock(b) | vial à 300 µl |
| 004 010 006 | Duffy(a) | vial à 300 µl |
| 004 010 007 | Duffy(b) | vial à 300 µl |
| 004 010 008 | Kell-Kp(b)-Js(a) | vial à 300 µl |
| 004 010 009 | Indian(b) | vial à 300 µl |
| 004 010 010 | JMH | vial à 300 µl |
| 004 010 011 | Cellano-Kp(b)-Js(a) | vial à 300 µl |
| 004 010 012 | Lutheran(a) | vial à 300 µl |
| 004 010 013 | Lutheran(b) | vial à 300 µl |
| 004 010 014 | Landsteiner-Wiener(a) | vial à 300 µl |
| 004 010 015 | Rodgers(a) | vial à 300 µl |
| 004 010 016 | Scianna1 | vial à 300 µl |
| 004 010 017 | Xg(a) | vial à 300 µl |
| 004 010 018 | Cartwright(a) | vial à 300 µl |
| 004 010 019 | rBGA Kn(a)/DACY | vial à 300 µl |
| 004 010 020 | rBGA YCAD | vial à 300 µl |
* serological tested antigens – all relevant antigens on recombinant proteins are listed in the IFU
Publications



