HLA Real-time PCR
HLA-FluoGene® – The real-time detection for the perfect match
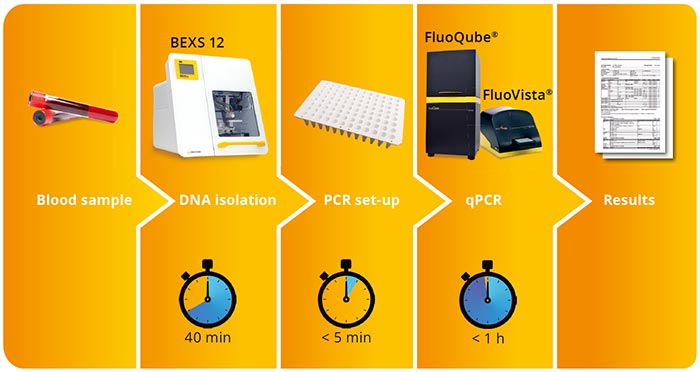
FluoGene® combines minimal hands-on time with reliable real-time PCR.

The advantages of the FluoGene® system
Established TaqMan® method.
Developed in close collaboration with histocompatibility and immunogenetics laboratories.
Low DNA consumption (e.g. 1 μg per 11 HLA loci with HLA-FluoGene Match).
Ready-to-use kits consisting of FluoMix, PCR plates with primer probes, and optical sealing foils.
Highest security due to barcoded tests and closed system.
Runs flexibly on the FluoVista® endpoint fluorescence reader and FluoQube® Real-time cycler.
User-friendly software with fully automated result calculation.
Same system incl. software for HLA, RBC, and HPA.
All products are CE IVD marked (outside USA & Canada) and are manufactured and tested under a DIN EN ISO 13485 certified quality system.
Downloads
Flyer HLA-FluoGene®
The next generation: HLA-FluoGeneNX Match and HLA-FluoGene384
Typing of 11 HLA loci with single antigen bead resolution: HLA-A, -B, -C, -DPA1, -DPB1, -DQA1, -DQB1, -DRB1, and -DRB3/4/5.
Typing of all 23 known CWD null alleles.
Hands-on time: less than 5 minutes.
Assay duration: less than 1 hour.
HLA-FluoGeneNX Match in 96 well format on the FluoQube®.
HLA-FluoGene384 Match in 384 well format on the FluoQube384 with 1 test (on half of PCR plate).
NEW: HLA-FluoGene384 Match in 384 well format on the FluoQube384 with 2 tests on a PCR plate.
Downloads
Flyer FluoGene®
Flyer FluoGene® NX Kits
RT-PCR Cycler for HLA-FluoGene Kits
RT-PCR Cycler for HLA-FluoGene384 Kits
Products:
HLA FluoGene Kits - 15 µl format
| Item number | Name | Test / Plate |
|---|---|---|
| 002 070 010 | HLA-FluoGene A | 1 |
| 002 072 010 | HLA-FluoGene C | 1 |
| 002 073 010 | HLA-FluoGene ABC | 1 |
| 002 077 010 | HLA-FluoGene DRDQ | 1 |
| 002 077 030 | HLA-FluoGene DRDQ | 3 |
| 002 078 048 | HLA-FluoGene B27 | 12 |
| 002 078 00Q | HLA-FluoGene B27 Q | 12 |
| 002 079 010 | HLA-FluoGene DPB1 | 1 |
| 002 081 010 | HLA-FluoGene DRDQDP plus | 1 |
| 002 084 020 | HLA-FluoGene DRDQ plus | 2 |
| 002 085 010 | HLA-FluoGene384 Match | 10 |
| 002 07W T96 | FluoGene Wipe Test | 2 |
HLA FluoGene NX Kits - 8 µl format
| Item number | Name | Test / Plate |
|---|---|---|
| 002 085 010 | HLA-FluoGene 384 Match | 1 |
| 002 085 020 | HLA-FluoGene 384 Match | 2 |
| 002 082 010 | HLA-FluoGeneNX ABCDRDQ | 1 |
| 002 083 005 | HLA-FluoGeneNX Match | 1 test on 2 plates |
| 002 074 010 | HLA-FluoGene ABDR | 1 |
| 002 071 020 | HLA-FluoGene B | 2 |
| 002 071 010 | HLA-FluoGene B | 1 |
| 002 073 010 | HLA-FluoGene ABC | 1 |
| 002 081 010 | HLA-FluoGene DRDQDP plus | 1 |
| 002 077 010 | HLA-FluoGene DRDQ | 1 |
| 002 077 030 | HLA-FluoGene DRDQ | 3 |
| 002 078 048 | HLA-FluoGene B27 | 12 |
Licensor Dyes and Quenchers
“HilyteTM” is a trademark of Anaspec, Inc., “QXL®” is a registered trademark of Anaspec, Inc., “DDQITM” are trademarks of Kaneka Eurogentec SA.
QXL, Hilyte, DDQI, DDQII, are licenced pursuant to an agreement with Kaneka Eurogentec S.A., and these products are sold exclusively for diagnostic purposes.
Elitech Dyes and Quenchers
“EclipseTM Dark Quencher”, “EclipseTM Quencher” are trademarks of ELITechGroup, Inc., “Yakima Yellow®”, “AquaPhluor®”, “Redmond Red®” and “Eclipse®” are registered trademarks of ELITechGroup, Inc.
Use of this product (including but not limited to Yakima Yellow® and EclipseTM Dark Quencher) is covered by patents owned by ELITechGroup, Inc., and sublicensed by Kaneka Eurogentec S.A. Such patents include without limitation the following US patents and corresponding patent claims outside the US: US6,699,975, US 6,790,945, US 7,662,942, US 6,653,473, US 6,972,339, 7,541,454, 7,671,218, 7,767,834, 8,163,910, 6,727,356. Further information on purchasing licences may be obtained by contacting licensing@eurogentec.com.
QXL, Hilyte, DDQI, DDQII, are licenced pursuant to an agreement with Kaneka Eurogentec S.A., and these products are sold exclusively for diagnostic purposes.
Elitech Dyes and Quenchers
“EclipseTM Dark Quencher”, “EclipseTM Quencher” are trademarks of ELITechGroup, Inc., “Yakima Yellow®”, “AquaPhluor®”, “Redmond Red®” and “Eclipse®” are registered trademarks of ELITechGroup, Inc.
Use of this product (including but not limited to Yakima Yellow® and EclipseTM Dark Quencher) is covered by patents owned by ELITechGroup, Inc., and sublicensed by Kaneka Eurogentec S.A. Such patents include without limitation the following US patents and corresponding patent claims outside the US: US6,699,975, US 6,790,945, US 7,662,942, US 6,653,473, US 6,972,339, 7,541,454, 7,671,218, 7,767,834, 8,163,910, 6,727,356. Further information on purchasing licences may be obtained by contacting licensing@eurogentec.com.
For USA and CANADA: Products are not FDA cleared devices and are for Research Use Only (RUO).


